Scientists have discovered that aminoglycoside antibiotics, a crucial class of drugs against severe bacterial infections, enter bacteria not through passive diffusion, but by hijacking sugar transporters. This breakthrough, published in Science Advances on September 5, 2025, by researchers at the Institut Pasteur and affiliated institutions, could revolutionize antibiotic treatment strategies, particularly in the face of rising antibiotic resistance.
The Long-Standing Mystery of Antibiotic Entry
For decades, how aminoglycosides breached bacterial defenses remained unclear. These antibiotics effectively kill bacteria like Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus by blocking protein synthesis. However, some strains, including increasingly resistant E. coli, have evolved ways to evade treatment, contributing to an estimated 829,000 deaths worldwide in 2019. The prevailing theory suggested aminoglycosides passively crossed the bacterial cell envelope, but new evidence proves this is not the case.
The Role of Sugar Transporters
Researchers initially stumbled upon this discovery while studying bacterial stress responses to antibiotics in Vibrio cholerae. They observed a correlation between antibiotic efficacy and the presence of sugar transporters – molecular “gateways” bacteria use to import essential carbohydrates like glucose, sucrose, and fructose. Intrigued, they investigated this mechanism in E. coli.
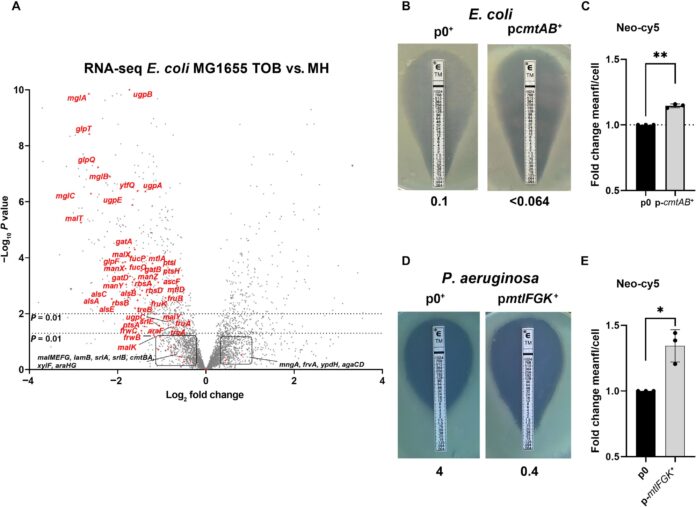
Using fluorescent aminoglycosides, they directly observed the antibiotics actively entering bacterial cells through these sugar transporters. This marks the first time an antibiotic has been proven to utilize this mode of transport. “It was an unexpected finding,” explains Zeynep Baharoglu, lead author and Research Director at the Institut Pasteur. “But the data was undeniable.”
Boosting Antibiotic Effectiveness with Uridine
The team realized that sugar transporters are not fixed; their numbers fluctuate based on the surrounding environment. By manipulating the availability of certain sugars, they could potentially increase transporter abundance and, consequently, antibiotic permeability.
After screening 200 compounds, they identified uridine as a particularly effective candidate. In both human biological samples contaminated with E. coli and animal models of urinary tract infection, uridine doubled the number of sugar transporters, leading to a tenfold increase in bacterial sensitivity to aminoglycosides. Remarkably, even drug-resistant and multidrug-resistant strains regained susceptibility when uridine was present.
Implications for Antibiotic Resistance and Future Treatments
This discovery has far-reaching implications. By administering uridine alongside aminoglycosides, doctors may be able to reduce antibiotic dosages, minimizing the risk of further resistance development and reducing toxic side effects. High doses of aminoglycosides can damage the inner ear and kidneys, a concern that lower doses could mitigate.
“This is a major discovery that could be a game changer for this antibiotic class,” says Baharoglu. “It could enable us to use lower concentrations or shorter treatments, extending the usefulness of these drugs.”
Another possibility is “grafting” uridine onto other antibiotics to enhance their entry into resistant bacteria. The fact that uridine is already used clinically and has demonstrated human safety could accelerate the development and testing of these new strategies.
A Critical Step in Combating Antibiotic Resistance
The World Health Organization estimates that antibiotic-resistant bacteria were linked to over 6 million deaths in 2019. This research underscores the importance of fundamental scientific inquiry in addressing this growing global threat. “Without this basic research, this discovery, which could play a key role in future strategies to combat antibiotic resistance, would not have been possible,” concludes Didier Mazel, Head of the Bacterial Genome Plasticity Unit at the Institut Pasteur. This breakthrough offers a renewed hope in the fight against antibiotic resistance, by unlocking a new mechanism to enhance the efficacy of existing drugs